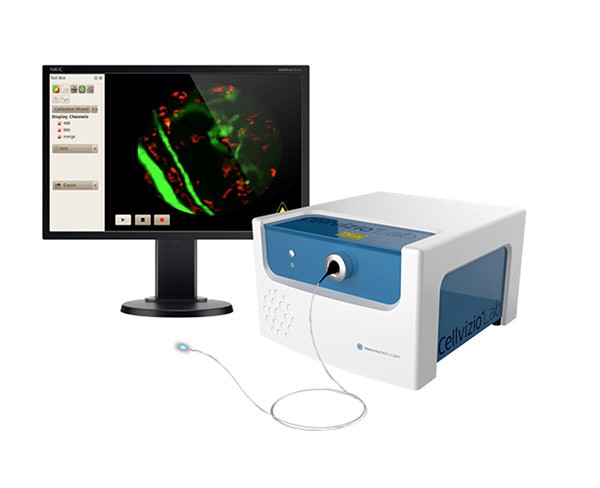
 Infrastruktura badawcza naszego Instytutu wzbogaciła się o wysokiej klasy aparaturę – System Cellvizio® Dual Band firmy Manua Kea Technologies służący do przyżyciowego obrazowania w badaniach przedklinicznych, w rozdzielczości komórkowej.
Infrastruktura badawcza naszego Instytutu wzbogaciła się o wysokiej klasy aparaturę – System Cellvizio® Dual Band firmy Manua Kea Technologies służący do przyżyciowego obrazowania w badaniach przedklinicznych, w rozdzielczości komórkowej.
System Cellvizio®, wykorzystujący technologię konfokalnej fluorescencyjnej endoskopii laserowej, jest jednym z dwóch tego typu urządzeń funkcjonujących obecnie w Polsce. Zastosowane rozwiązania pozwalają na wykonywanie bezpośredniego obrazowania tkanek i organizmów w czasie rzeczywistym metodami nieinwazyjnymi lub minimalnie inwazyjnymi.
System stanowi połączenie mikroskopu konfokalnego z giętkim endoskopem, czerpiąc korzyści z obu tych technologii. Dzięki czujnikowi endoskopowemu można prowadzić zarówno obrazowanie powierzchniowe (przyłożenie czujnika do powierzchni badanej tkanki), jak i wewnętrzne (wkłucie czujnika w miękką tkankę). Z kolei dostępność obrazowania konfokalnego pozwala na uzyskanie rozdzielczości na poziomie komórkowym, niedostępnej w innych systemach do obrazowania in vivo (m.in. MRI, CT, PET, SPECT, USG)
Wśród korzyści płynących z zastosowania endo-mikroskopii, in vivo, w czasie rzeczywistym, na uwagę zasługują:
• dostęp eksperymentatora do dowolnego miejsca u badanego zwierzęcia w trakcie całego procesu obrazowania
• możliwość przeprowadzenia badań morfologicznych i czynnościowych zarówno u zwierzęcia czuwającego po uprzednim unieruchomieniu, jak i u zwierzęcia w narkozie wziewnej (lekka anestezja)
• możliwość śledzenia zmian u tego samego zwierzęcia przez długi okres czasu in vivo
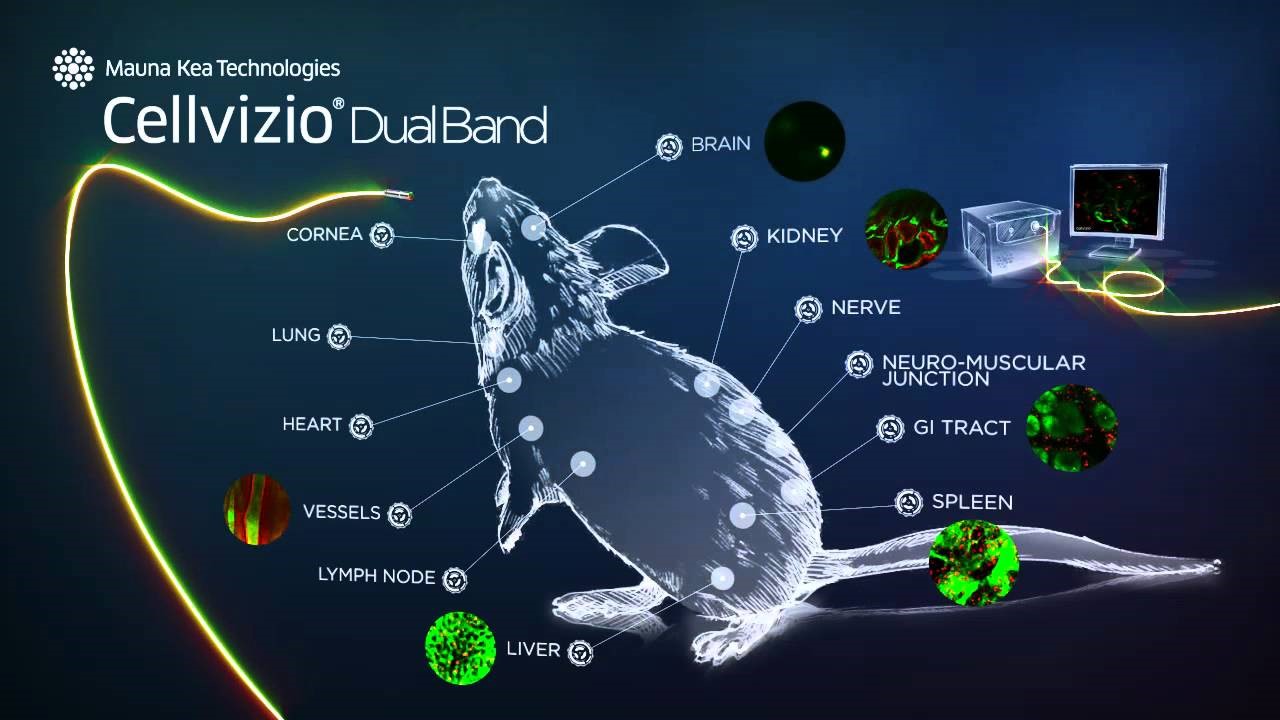
Cechy te umożliwiają prowadzenie szerokiego spektrum badań, w tym:
• badania unaczynienia – jego gęstości, morfologii i czynności
• dynamiczne śledzenie interakcji komórkowych i molekularnych
• badanie biodystrybucji leków, substancji endo- i parakrynnych
• monitorowanie zmian fizjologicznych i funkcjonalnych w kontekście anatomicznym
System Cellvizio® IMDiK PAN usytuowany jest obecnie w Zakładzie Fizjologii Nerek i Płynów Ustrojowych, pod nadzorem Prof. dr hab. Elżbiety Kompanowskiej-Jezierskiej:
tel. 22 60 86 546; E-mail: This email address is being protected from spambots. You need JavaScript enabled to view it.
Zapraszamy wszystkich zainteresowanych użytkowników wewnętrznych i zewnętrznych do kontaktu z opiekunem aparatury.
Ponadto uprzejmie informujemy, że poszukujemy obecnie operatora urządzenia – osoby chętnej do przeszkolenia przez producenta, która będzie następnie odpowiedzialna za jego obsługę, opracowanie protokołów barwień poszczególnych organów i tkanek, przygotowywanie materiału badawczego, wykonywanie obrazowania, dekonwolucję i analizę obrazów konfokalnych.
Zainteresowane osoby prosimy o zgłaszanie się do opiekuna aparatury.






